After screening 12,000 drugs from the library of collections ReFRAME, scientists have isolated 21 drugs for their antiviral activity as effective in providing treatment to Covid-19 patients depending upon dosage and other modifications. The list includes astemizole for allergies and clofazamine for leprosy, and remdesivir, which are already approved by the the U.S. Food and Drug Administration (FDA).
A Nature study authored by a global team of scientists led by Sumit Chanda, professor at Sanford Burnham Prebys Medical Discovery Institute, has identified these existing drugs that stop the replication of SARS-CoV-2, which causes coronavirus or COVID-19.
The scientists analyzed 12,000 known drugs for their ability to block the replication of SARS-CoV-2, and reported 100 molecules with confirmed antiviral activity in tests and found 21 of these drugs to be effective at concentrations that could be safely given to patients. Moreover, four of these compounds were found to work synergistically with remdesivir, which is now a standard-of-care treatment for COVID-19 patients.
“Remdesivir has proven successful at shortening the recovery time for patients in the hospital, but the drug doesn’t work for everyone who receives it. That’s not good enough,” says Chanda. “As infection rates continue to rise in America and around the world, the urgency remains to find affordable, effective, and readily available drugs that can complement the use of remdesivir, as well as drugs that could be given prophylactically or at the first sign of infection on an outpatient basis.”
The team conducted extensive testing and validation to evaluate the drugs on human lung biopsies of Covid-19 patients and also evaluated the drugs for synergies with remdesivir, besides monitoring dose-response relationship between the drugs and antiviral activity. Here’s the full list of 21 drugs:
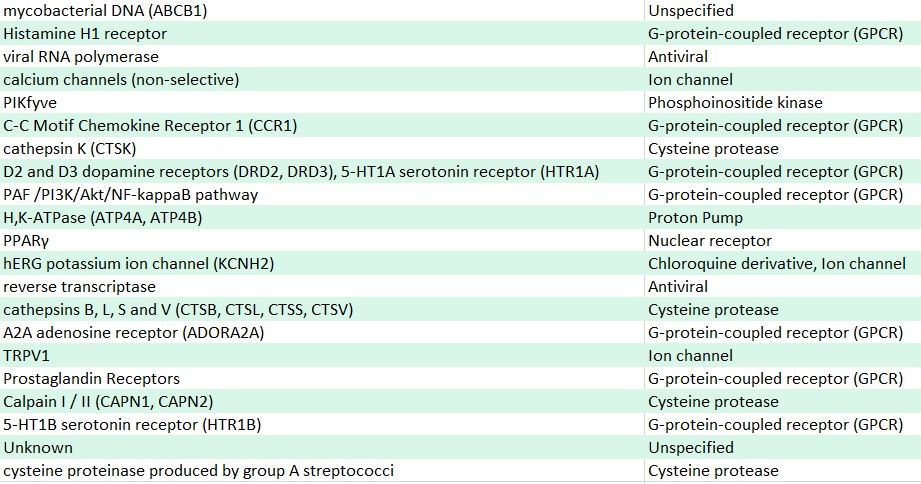
Of the 21 drugs,the scientists found:
- 13 have previously entered clinical trials and are found effective at concentrations, or doses, that could potentially be safe for COVID-19 patients.
- Two are already FDA approved — astemizole (allergies), clofazamine (leprosy), while remdesivir has received Emergency Use Authorization from the FDA.
- Four worked synergistically with remdesivir, including the chloroquine derivative hanfangchin A (tetrandrine), an antimalarial drug that is into Phase 3 clinical trials.
What’s Next?
The researchers are currently testing all 21 compounds in small animal models and lung organoids that mimic human tissue. Once these studies are favorable, the team hopes to approach the FDA for more clinical trials to test these drugs for COVID-19 patients.
The drugs were identified after screening of more than 12,000 drugs from the ReFRAME drug repurposing collection–the most comprehensive drug repurposing collection of compounds that have been approved by the FDA for other diseases.