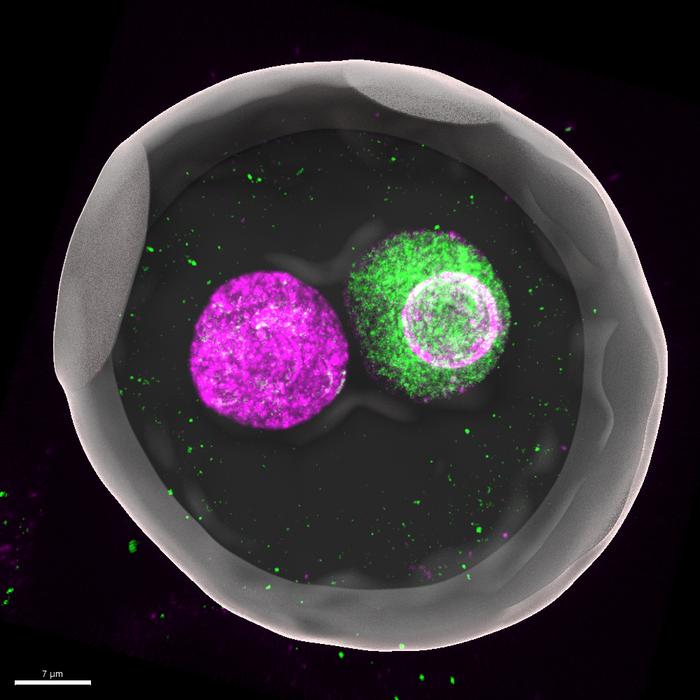
The sperm and the egg cell’s nuclei compete for size directly after fertilization and this is necessary for the proper embryonic development. The mouse study with Kobe University participation finally gives meaning to a phenomenon biologists have known for decades.
At the beginning of a new individual’s life, an egg and a sperm fuse to form what biologists call a “zygote,” a fertilized egg. In mammals such as mice and humans, the DNA-carrying nuclei of these cells don’t fuse right away but remain separate as so-called “pronuclei” until just before the first cell division. “This fact has been known for decades, but no one really understood why this separation exists. People just treated it as a descriptive feature rather than something to be tested experimentally,” says Kobe University developmental biologist KYOGOKU Hirohisa. If anything, separate nuclei may increase the risk of errors during the first cell division. In assisted reproduction, embryos in which the maternal and paternal pronuclei are fused early have therefore attracted interest, but these embryos are known to have a lower likelihood of developing to term.
Kyogoku specializes in studying early development directly after fertilization. “The DNA an individual inherits from their mother and father differs not only in its genetic information. It is also known that the maternal genome has a lot of chemical modifications, like little tags, on the molecules around which the DNA is wrapped. The genome from the sperm, on the other hand, has virtually no such modifications. But these proper modifications are essential for embryonic development,” he explains. Thus suspecting that the fusion of pronuclei might have an influence on gene regulation, he teamed up with researchers from RIKEN and, using his special cell manipulation techniques, took a close look at what happened to these modifications when the pronuclei are fused and under other conditions.
In the journal Nature, the team now published that they found that the size of the pronuclei is essential for maintaining the regulatory modifications. Whenever the size of the nucleus was large, either through manipulation or because of the premature fusing of the maternal and paternal pronuclei, the degree of the chemical tagging became low. And when they investigated what keeps the pronuclei’s sizes small in the natural, separated state, they found that the two pronuclei engage in a race to absorb factors that regulate nucleus growth. This then yielded a clear mechanistic explanation for why the developmental potential of fused nuclei is smaller: Because the single nucleus doesn’t have to compete for these factors it ends up much larger and as a result the genome’s regulatory tags get lost. “I find it exciting that a very familiar structure — two separate nuclei in a fertilized egg — turns out to have a clear and functional role, that is, it actively creates a competitive environment inside the cell which helps maintain proper regulation and supports development,” says Kyogoku.
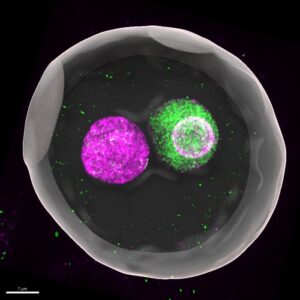
In mammals such as mice and humans, the DNA-carrying nuclei of these cells don’t fuse right away but remain separate as so-called “pronuclei” until just before the first cell division. This 3D-reconstructed image shows a mouse zygote (fertilized egg), where maternal (magenta) and paternal (green) genomes are enclosed in separate pronuclei. “This fact has been known for decades, but no one really understood why this separation exists,” says Kobe University developmental biologist KYOGOKU Hirohisa. Credit: KYOGOKU Hirohisa
To test their theory, the team temporarily introduced into zygotes with prematurely fused pronuclei an additional pronucleus to re-establish competition. And indeed, this intervention successfully limited nuclear size and partially restored the regulatory marks as well as the developmental potential.
The Kobe University work opens a whole new chapter of research into the very first steps a new life takes. “Even at the beginning of life, spatial organization is not just incidental but fundamentally important,” explains Kobe University biologist Kyogoku. He continues, “This result brings us one step closer to understanding the physical and biological principles that explain why early embryos are both robust and error-prone at the same time.”
Also Read:
Scientists Develop Smart Oxide That Stores Power And Shows Charge Status
Primary Breast Tumours Found To Already Contain Cells With Metastatic Potential